
Introduction
過去,醫學界對於相同癌症患者治療效果差異的原因缺乏深刻了解,而腫瘤內的異質性被證明是其中的關鍵。腫瘤內存在基因組變異、結構變化、染色質修飾的異質性,使癌症治療變得複雜。治療標的變化與異質性增加及基因表達的變化密切相關,對癌症治療產生直接影響。
In the past, the field of medicine lacked a profound understanding of the reasons for variations in treatment outcomes among patients with the same cancer, with tumor heterogeneity proving to be a key factor. Heterogeneity within tumors, encompassing genomic variations, structural changes, and chromatin modifications, complicates cancer therapy. Changes in treatment targets are closely associated with increased heterogeneity and alterations in gene expression, directly impacting cancer treatment.
為了克服治療中的挑戰,我們提倡以空間體學的角度進行解析,尋找癌症治療的新可能性。空間體學為現今的熱門顯學,解決過往樣品老舊完全無法檢測、分辨力不足等難題,開發出具革命性的空間體學技術平台-整合了單細胞成像、流體力學、化學和生物資訊學等技術,打破以往僅能藉由染色得知有限的蛋白表現,組織內細胞間彼此的交互作用能被直接得知。針對各樣疾病的標定不再是模糊不清或是僅能依靠有限資訊推敲可能治療策略。空間體學有助於深入理解細胞的位置,以及其在疾病發展和藥物反應中的交互作用,為轉譯醫學帶來洞察力驅動的革新研究!
To overcome the challenges in treatment, we advocate for an analysis from a spatial omics perspective to explore new possibilities in cancer therapy. Spatial omics is currently a prominent field addressing past difficulties such as the inability to detect old samples, as well as insufficient resolution, by developing revolutionary spatial omics technology platforms. These platforms integrate techniques such as single-cell imaging, fluid dynamics, chemistry, and bioinformatics, breaking the limitations of only being able to ascertain limited protein expression through staining. The interactions between cells within tissues can now be directly understood. Targeting various diseases is no longer vague or reliant solely on limited information to infer possible treatment strategies. Spatial omics aids in understanding the precise location of cells and their interactions in disease progression and drug response, driving insight-driven innovative research in translational medicine!
|
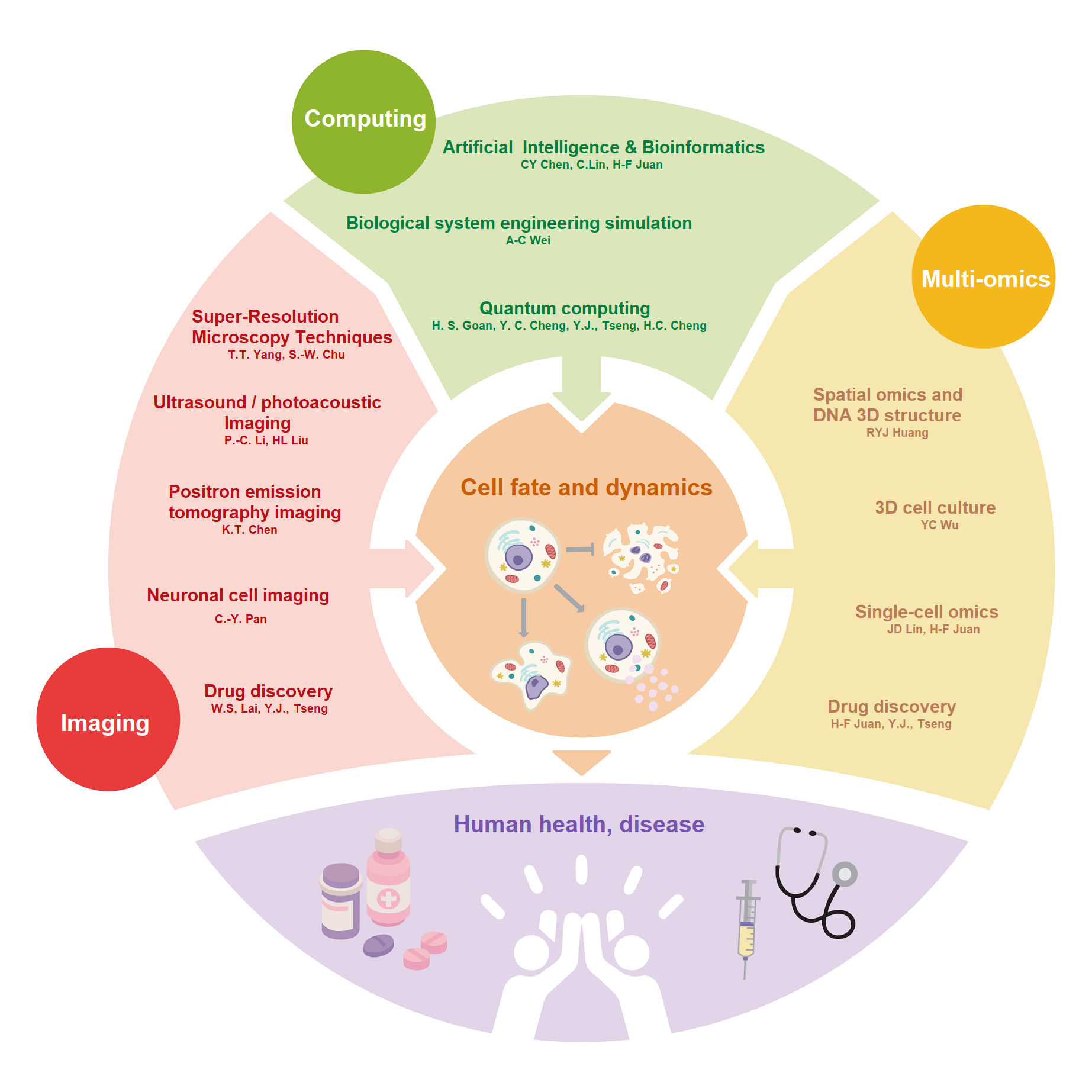
在尖端生醫計算及影像研究中心(CACIB),計算、影像與多體學是三大主題。這些領域的發展有助於實現精準健康和醫療。結合多體學、計算科學與多模態影像技術,有望更深入了解致病機轉,解決難以應對的疾病,促進新藥開發的進展,為實現個人化醫療帶來曙光。 |

|
|
At the Center of Advanced Computing and Imaging in Biomedicine (CACIB), computation, imaging, and multi-omics are the three main themes. The development in these areas contributes to achieving precision health and medicine. By combining multi-omics, computational science, and multimodal imaging techniques, we hope to gain a deeper understanding of pathogenesis, address challenging diseases, promote advancements in drug development, and shed light on personalized medicine. |
|
Agenda
會議議程的安排以「空間體學技術的創新」為開場,並邀請技術開發的專家學者們,圍繞著「我們是如何走到現在的?」的主題展開討論。 第二天(5/24)則有4大主題的討論:首先聚焦在「空間體學技術在腫瘤異質性中的應用」,並深入研究相關領域的最新發展。接下來在討論「空間體學的突破」時,將聚焦於該領域的技術創新和應用前景,以及相關研究的未來方向。下午的會議將引導參與者「運用空間體學的思維在研究設計中的應用」,促使與會者更深入地思考和應用這一技術。 當天最後的議題將聚焦於如何「利用空間體學技術改變病理學」。這項討論將關注技術在病理學研究和診斷中的創新應用,以及這些應用如何推動醫學領域的進步。 最後一天(5/25)的討論將集中在「多體學的未來」。並邀請技術專家、藥廠研發人員以及腫瘤科醫師參與。各自從技術開發者、藥廠生物標誌的開發、以及臨床學家的角度,他們將分享對空間體學技術的看法,促使多方面的討論,從而更全面地理解和應用這一前沿技術。